|
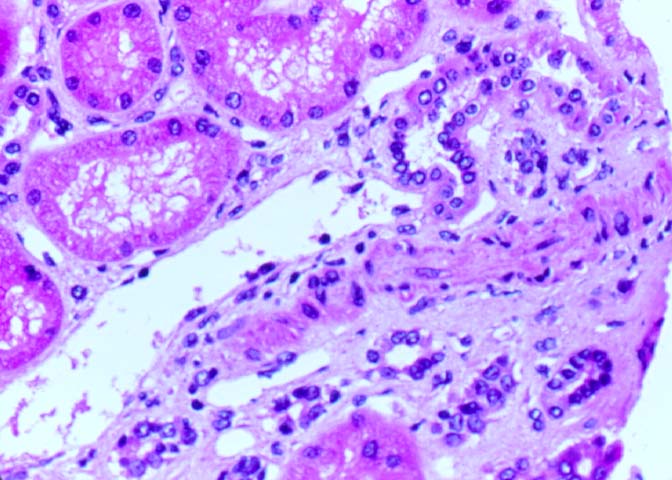
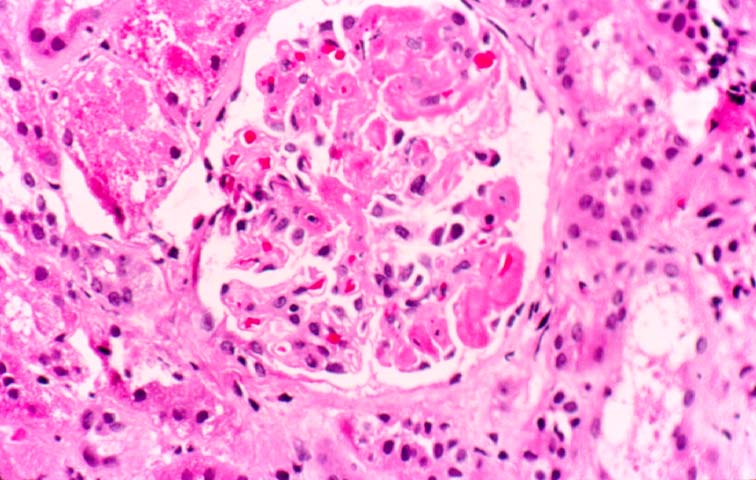
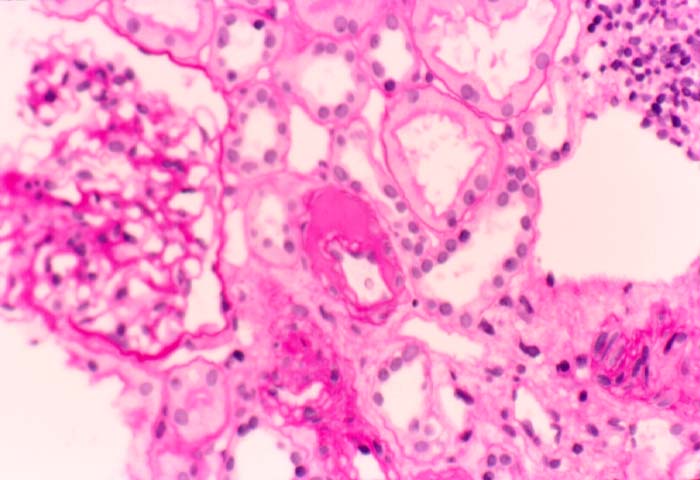
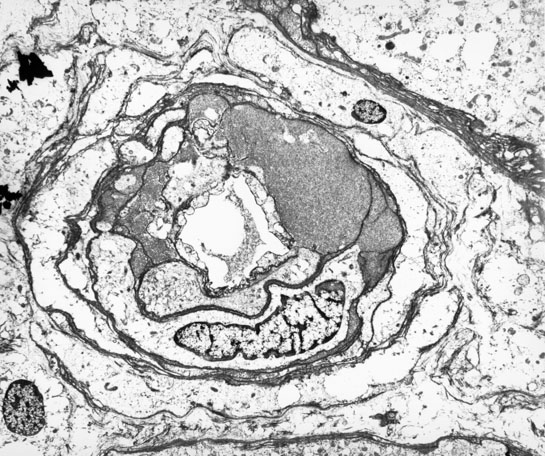
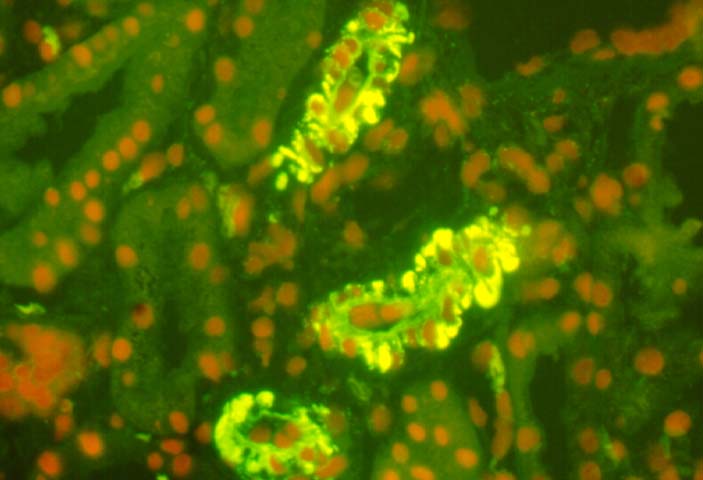
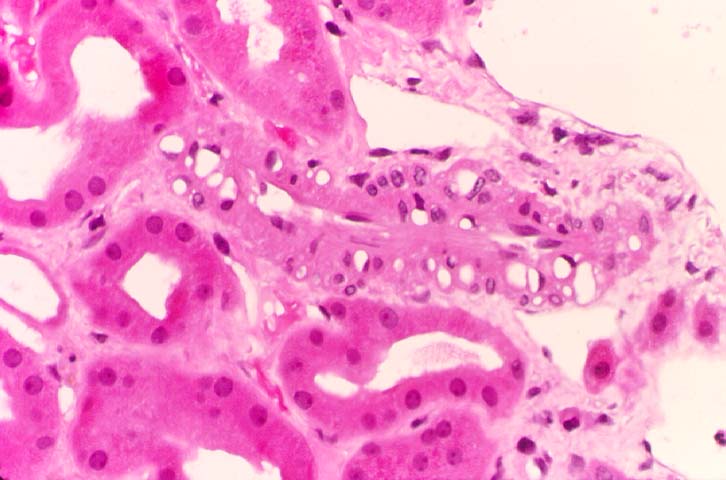
Microvascular Toxicity Cyclosporine and tacrolimus can damage the glomerular capillaries and renal arterioles. Thus allografts maintained on cyclosporine show scattered thrombi in the glomerular capillaries and arterioles in 3-26% of biopsy samples obtained in the first week post-transplant. Clinically, the patients may be asymptomatic, but when the changes are marked, hemolytic uremic syndrome can supervene. An acute arteriolopathy involving small vessels less than 2 smooth muscle cells in thickness has been linked to cyclosporine therapy, and we have seen a few cases with tacrolimus as well. The injury is characterized by endothelial swelling, and mucoid intimal thickening. Differentiating the lesion from vascular rejection is critical: the presence of significant lymphocytic infiltrates in the intima, scattered interstitial hemorrhages and tubulitis should strongly favor the diagnosis of rejection over drug toxicity. Cyclosporine and tacrolimus therapy can both result in arteriolar hyaline eosinophilic deposits comprised of fibrin, IgM, C3, and C1q. These deposits are particularly seen after prolonged drug therapy, but can conceivably develop rapidly after acute arteriolopathy. A non-specific form of hyaline change can occur with ageing, hypertension and diabetes mellitus. On the other hand, hyalin with a nodular configuration to the hyalin has been considered to be specific for cyclosporine toxicity. However, nodular hyalinosis has become uncommon with currently used doses of cyclosporine. Moreover, this change has also been described in donor transmitted nephrosclerosis, and in patients dying of ischaemic cardiomyopathy. The natural history of arteriolar hyalinosis has not been well investigated. Uncontrolled studies on cyclosporine treated patients suggest that discontinuation of the drug is followed by resolution of hyalin deposition in some cases, but continued progression in others. Evaluation of such data is complicated by the extremely focal nature of these lesions, which may may involve less than 10% of vessels sampled at biopsy. Arteriolar myocyte vacuolization can accompany acute arteriolopathy as well as arteriolar hyalinosis. This lesion is a non-specific manifestation of vessel spasm, and should be attributed to drug toxicity only after exclusion of other causes of vascular injury. Thus, myocyte vacuolization can be seen in moderate to severe rejection with intimal arteritis, and in native kidneys with arteriolonephrosclerosis. Amphotericin B can also cause striking smooth muscle cell vacuolization in the media of renal arterioles and small arteries.
Please mail comments, corrections or suggestions to the TPIS administration at the UPMC.
If you have questions, please email TPIS Administration. |
||||